General scope
Our research is focused on bioelectrical phenomena and, in particular, on exploring the use of these phenomena for developing new methods and devices for biomedical applications.
Our research is focused on bioelectrical phenomena and, in particular, on exploring the use of these phenomena for developing new methods and devices for biomedical applications.
Since 2011, we have been pioneering a novel approach to developing injectable electronic devices for therapeutic and diagnostic applications, from neuroprosthetics to remote patient monitoring. Visit our eAXON endeavour website to know more about it.
Electroporation, or electropermeabilization, is the phenomenon by which cell membrane permeability to ions and macromolecules is increased by exposing the cell to short high electric field pulses.
Electroporation is a dynamic phenomenon that depends on the local transmembrane voltage. It is generally accepted that, for a given pulse duration and shape, a specific transmembrane voltage threshold exists for the manifestation of the electroporation phenomenon (from 0.2 V to 1 V). This leads to the definition of an electric field magnitude threshold for electroporation (Erev); only the cells within areas where the electric field magnitude is larger than Erev are electroporated. If a second threshold (Eirrev) is reached or surpassed, the electroporation phenomenon will be too intense and cell homeostasis will be altered to the point of compromising cell viability which can finally result in cell death, either necrotic or apoptotic.
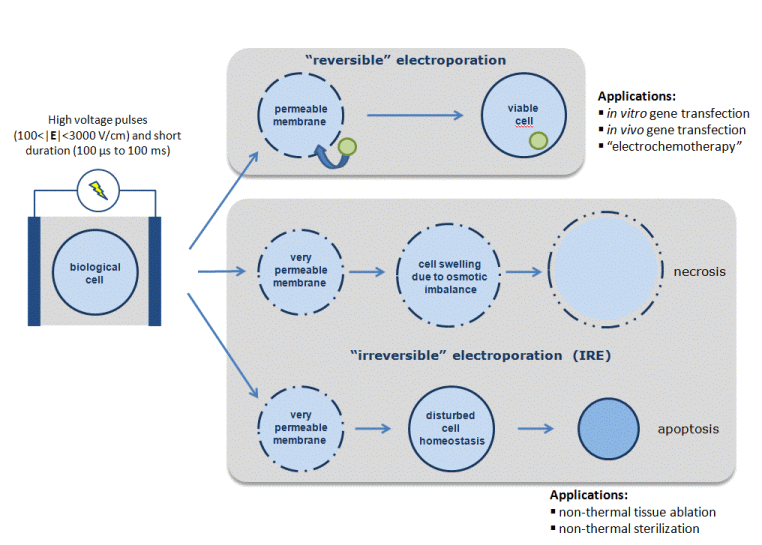
Reversible electroporation of living tissues is the basis for different therapeutic maneuvers on clinical use such as the in vivo introduction of genes into cells ("electrogenetherapy") and the introduction of anti-cancer drugs into cells of solid tumors ("electrochemotherapy"). Recently, irreversible electroporation has also found a use in tissues as a minimally invasive surgical procedure to ablate undesirable tissue with important advantages when compared to thermal ablation techniques.
A specific objective of our research is to develop new electrode arrangements, devices and methodologies for electric field management in electroporation therapies.

BERG has two electronics laboratories (30 m2 + 30 m2) with state-of-the-art equipment for the development and testing of microdevices and electronic systems. Among other specialized equipment, we have a high precision welding laser system, a coiling machine, a wire bonding machine for die assembly, high-end inspection microscopes, a rework station, a thermal imaging camera, and equipment for electronic testing (oscilloscopes, function generators, active differential probes, etc.). For prototyping, an air compressor with a silicone dispenser, a vacuum chamber, 3D printers, and a CNC machine are available. Workstations and software licenses (Cadence, COMSOL Multiphysics + modules, MATLAB, etc.), are available for numerical modeling and for the design of electronic systems and integrated circuits. At the Barcelona Biomedical Research Park (PRBB) we have two wet laboratories (10 m2 + 20 m2) equipped for conventional microbiology and we have access to PRBB scientific services.